Three Things You Should Know About LC-MS Analysis
Commonly used in food science, pharmaceuticals, and forensic toxicology, the instrument liquid chromatography-mass spectrometry (LC-MS) can identify most organic compounds ranging from proteins to pharmaceutical products in the market.
Compared to the analytical technique GC-MS, LC-MS has a wider usage to analyze chemical compounds that are thermally unstable, non-volatile, polar, ionic, and large. The improvements that transcend LC-MS to become the easy-to-use, selective and sensitive technique it is today have made the instrument more appealing in various applications.
What Is LC-MS?
LC mass spectrometry is an analytical technique that combines chromatographic separation of target compounds (or analytes) followed by their mass-based detection. The sensitivity, selectivity, and accuracy especially of liquid chromatography-tandem triple-quadrupole mass spectrometry (LC-MS) have resulted in widely utilized procedures optimal for detecting nanomolar or even picomolar quantities of various analytes ranging from drugs, drug and food metabolites, biomarkers of disease progression or drug efficacy, pesticides, food contaminants, markers of ecosystem stability, and natural product extracts.
How does LC-MS Work?
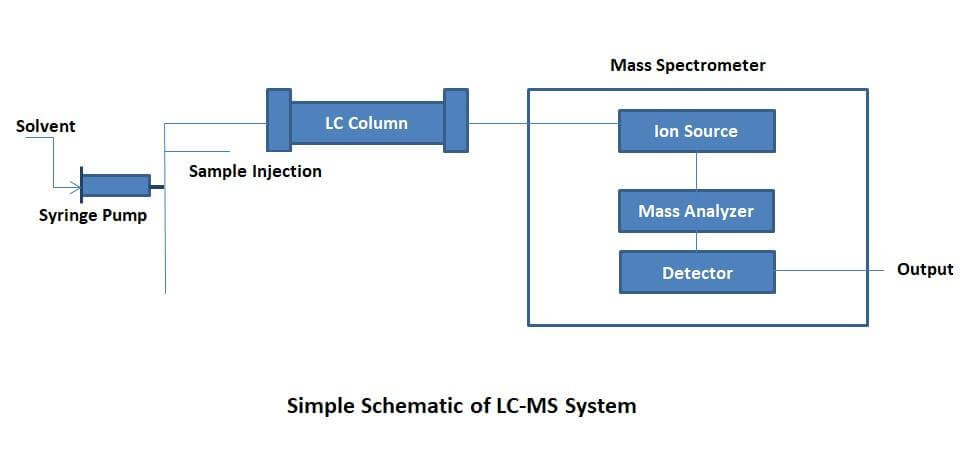
LC-MS involves separating mixtures in accordance with their physical and chemical properties, then identifying the components within each peak and detecting them based on their mass spectrum. The flow rates used in LC-MS should be less than those used for HPLC.
This is to ensure complete ionization and to maintain the detection sensitivity of the MS, which starts to decrease beyond 200 µL/min. Therefore, the column in LC-MS is much smaller to accommodate the smaller solvent flow rates and sample volumes.
This makes syringe pumps very convenient for LC-MS because they are very accurate and can deliver very low flow rates. In addition, it is possible to use syringe pumps for sample injection into the system as they can deliver very precise sample dosing.
The Applications of LC-MS
LC-MS is a common technique in forensic laboratories to identify various illicit drugs in biological as well as chemical matrices, dyes in fibers as trace evidence, and fire debris from a particular crime scene.
More commonly, approximately 70% of the samples brought to forensic toxicology laboratories are handled by liquid chromatography for their efficiency in detecting polar and thermally labile chemical compounds. The instrument’s ability to analyze semi-volatile samples, its rapidness, and the lack of derivatization needed, have made LC-MS the more favorable technique compared to GC-MS.
Applications of the instrument within the pharmaceutical industry involve the analysis of suspected drug substances occurring in natural and synthetic sources, as well as drug metabolism in the body. The instrument has allowed drug manufacturers to develop, compare, and control the quality of drug products through its chromatography element. Its mass spectrometry element serves to improve the limits of detection for pharmaceutical analysis of complex drug mixtures, as well as their molecular structures.
The use of LC-MS has also been growing rapidly in drug development over many years. Its improvements in its features to quantify drugs, as well as qualitative and structural analysis of drugs, has provided the pharmaceutical industry with more advancements. It further provides a rapid turnaround of samples without compromising the quality of the analysis to generate accurate information, which is a primary advantage for the fast-growing pharmaceutical industry.
In environmental sciences, LC-MS is useful for the identification and quantification of compounds in drinking water, wastewater, and soil. For instance, the class of pesticides carbamates can be detected by the APCI-LC/MS without derivatization, compared to the common method of post-column derivatization and fluorescence detection.
Through APCI-LC/MS, the individual chemical components in a matrix can be detected through the corresponding peaks on an extracted ion chromatogram without derivatization.
Back to List
-
 下午4:09Weighing the Pros and Cons of PTFE/Silicone Septa
下午4:09Weighing the Pros and Cons of PTFE/Silicone Septa -
 下午4:05Decoding Vial Discard Guidelines: Ensuring Precision in Chromatography
下午4:05Decoding Vial Discard Guidelines: Ensuring Precision in Chromatography -
 下午5:01Navigating Micro Inserts for HPLC Vials: A Comprehensive Guide
下午5:01Navigating Micro Inserts for HPLC Vials: A Comprehensive Guide -
.jpg) 下午2:02Common faults and solutions of automatic samplers(2)
下午2:02Common faults and solutions of automatic samplers(2) -
 下午5:08Ensuring Sample Integrity: Navigating EPA Storage Vials Stability Guidelines
下午5:08Ensuring Sample Integrity: Navigating EPA Storage Vials Stability Guidelines

